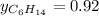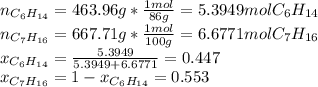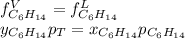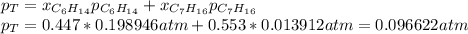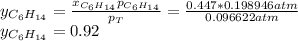Chemistry, 15.04.2020 00:51 malachilaurenc
Two linear hydrocarbons, Hexane (C6H14) and Heptane (C7H16), form pretty much an ideal solution at any composition. A solution is made at 25°C that contains 463.96 g of Hexane in 667.71 g Heptane: Characterise the vapour above this solution, and answer, What is the mole fraction of Hexane in the vapour?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 23.06.2019 02:10
Detrimental the length of the object shown 1. 97.8mm 2. 97.80 mm 3. 97mm 4. 98mm
Answers: 2

You know the right answer?
Two linear hydrocarbons, Hexane (C6H14) and Heptane (C7H16), form pretty much an ideal solution at a...
Questions








English, 29.08.2020 03:01




Social Studies, 29.08.2020 03:01



Physics, 29.08.2020 03:01




Mathematics, 29.08.2020 03:01