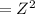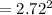Chemistry, 15.04.2020 01:53 sheldonwaid7958
Suppose you are studying the K sp of K C l O 3 , which has a molar mass of 122.5 g/mol, at multiple temperatures. You dissolve 4.00 g of K C l O 3 in 12 mL of water at 85 oC and cool the solution. At 74 oC, a solid begins to appear. What is the K sp of K C l O 3 at 74 oC?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:00
What postulate of the kinetic molecular theory best explains why gases have high fluidity? because collisions between gas particles are elastic, there is no loss of energy as particles flow past each other. because gases consist of large numbers of tiny particles, they spread out and do not come in contact with each other. because the attractive forces between gas particles are negligible, gas particles can glide easily past one another. because the average kinetic energy of gas particles increases as temperature increases, gas particles behave more like a liquid. question 6 compare the compressibility of gases and liquids. support your answer by describing the arrangement of particles in gases and liquids.
Answers: 1

Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2
You know the right answer?
Suppose you are studying the K sp of K C l O 3 , which has a molar mass of 122.5 g/mol, at multiple...
Questions





Chemistry, 17.07.2019 15:40

Mathematics, 17.07.2019 15:40

Chemistry, 17.07.2019 15:40

History, 17.07.2019 15:40

Mathematics, 17.07.2019 15:40

Biology, 17.07.2019 15:40

Physics, 17.07.2019 15:40

Mathematics, 17.07.2019 15:40

History, 17.07.2019 15:40

Mathematics, 17.07.2019 15:40


Chemistry, 17.07.2019 15:40



Social Studies, 17.07.2019 15:40

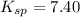
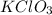 is = 122.5 g/ mol
is = 122.5 g/ mol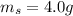
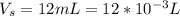
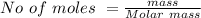
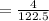
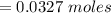
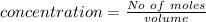
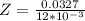
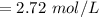
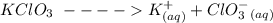
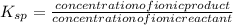
![K_{sp} = [k^+] [ClO_3^-]](/tpl/images/0600/6827/4dc14.png)