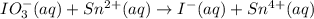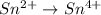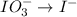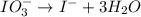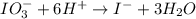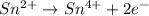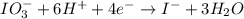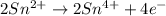Given the partial equation:
IO³⁻ (aq) + Sn²⁺ (aq) → I⁻ (aq) + Sn⁴⁺ (aq),
balance the reaction in acidic solution using the half-reaction method and fill in the coefficients.
The missing blanks represent H2O, H+, or OH-, as required to balance the reaction. Enter the coefficients as integers, using the lowest whole numbers. If the coefficient for something is "1", make sure to type that in and not leave it blank. Enter only the coefficients.
IO³⁻ (aq) + Sn²⁺ (aq) + → I⁻ (aq) + Sn⁴⁺ (aq) +

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2


Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

You know the right answer?
Given the partial equation:
IO³⁻ (aq) + Sn²⁺ (aq) → I⁻ (aq) + Sn⁴⁺ (aq),
balance the r...
IO³⁻ (aq) + Sn²⁺ (aq) → I⁻ (aq) + Sn⁴⁺ (aq),
balance the r...
Questions

Mathematics, 09.07.2019 22:00

Mathematics, 09.07.2019 22:00

Mathematics, 09.07.2019 22:00

Mathematics, 09.07.2019 22:00

Mathematics, 09.07.2019 22:00

Physics, 09.07.2019 22:00

Mathematics, 09.07.2019 22:00

Chemistry, 09.07.2019 22:00

Spanish, 09.07.2019 22:00


Mathematics, 09.07.2019 22:00


English, 09.07.2019 22:00


Physics, 09.07.2019 22:00

Mathematics, 09.07.2019 22:00

Chemistry, 09.07.2019 22:00

Mathematics, 09.07.2019 22:00


Mathematics, 09.07.2019 22:00

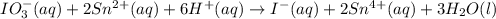
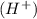 at that side where the less number of hydrogen are present.
at that side where the less number of hydrogen are present.