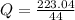Chemistry, 15.04.2020 02:11 espinosajoselyn
A 5.91 g of octane, C8H18, reacts with excess oxygen in a bomb calorimeter. The heat capacity of the calorimeter is 6.97 kJ/°C and the temperature increases by 32.0°C. How much heat, in units of kJ/mol, was absorbed by the bomb calorimeter?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 05:30
What is the mass of each element in a 324.8 sample of co2
Answers: 1

Chemistry, 22.06.2019 05:50
What are the 4 phases of matter in order of increasing engery content?
Answers: 2

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1
You know the right answer?
A 5.91 g of octane, C8H18, reacts with excess oxygen in a bomb calorimeter. The heat capacity of the...
Questions


Mathematics, 18.07.2019 03:30



Biology, 18.07.2019 03:30

Social Studies, 18.07.2019 03:30

Social Studies, 18.07.2019 03:30



Social Studies, 18.07.2019 03:30



Social Studies, 18.07.2019 03:30

Biology, 18.07.2019 03:30

Social Studies, 18.07.2019 03:30

Social Studies, 18.07.2019 03:30


Social Studies, 18.07.2019 03:30

Social Studies, 18.07.2019 03:30

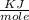
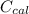 = 6.97
= 6.97 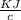
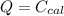 ΔT
ΔT