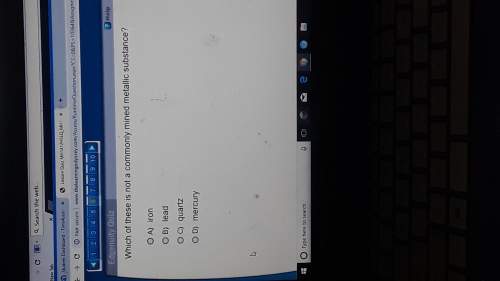
Chemistry, 15.04.2020 02:12 honeysenpai6
A student performed a serial dilution on a stock solution of 1.33 M NaOH. A 1.0 mL aliquot of the stock NaOH (ms) was added to 9 mL of water to make the first dilution (m1). Next, 1.0 mL of the m1 solution was added to 9 mL of water to make the second solution (m2). The processes was repeated for a total of 5 times. What is the final concentration of NaOH (m5)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:10
When electrolyzing copper (ll) chloride, what reaction takes place at the anode? what reaction takes place at the cathode?
Answers: 1

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1

Chemistry, 23.06.2019 05:00
Which characteristics affect ocean water’s temperature? check all that apply. depth location mass salinity waves
Answers: 1

Chemistry, 23.06.2019 06:00
In an exothermic reaction at equilibrium, what is the effect of lowering the temperature? a. the reaction makes more products. b. the reaction makes more reactants. c. the reaction is unchanged.
Answers: 1
You know the right answer?
A student performed a serial dilution on a stock solution of 1.33 M NaOH. A 1.0 mL aliquot of the st...
Questions

Mathematics, 21.05.2020 03:10




Mathematics, 21.05.2020 03:10

Mathematics, 21.05.2020 03:10


Mathematics, 21.05.2020 03:10

Mathematics, 21.05.2020 03:10


Physics, 21.05.2020 03:10

Mathematics, 21.05.2020 03:10


Biology, 21.05.2020 03:10


Biology, 21.05.2020 03:10



History, 21.05.2020 03:10