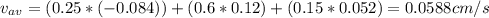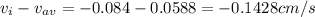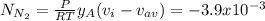A gaseous mixture at 265K and 1.0 atm contains 25 O2 60 N2 and 15 CO2 mole basis The velocities of the components are 0.084 cm s O2 0.120 cm s N2 and 0.052 cm s CO2 Find the N2 diffusion velocity relative to the mole average velocity and the molar diffusional flux of N2

Answers: 2
Another question on Chemistry




Chemistry, 22.06.2019 15:00
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2
You know the right answer?
A gaseous mixture at 265K and 1.0 atm contains 25 O2 60 N2 and 15 CO2 mole basis The velocities of t...
Questions

Chemistry, 24.04.2020 18:07

Mathematics, 24.04.2020 18:08

World Languages, 24.04.2020 18:08

Health, 24.04.2020 18:08





History, 24.04.2020 18:08



Mathematics, 24.04.2020 18:08



Social Studies, 24.04.2020 18:08

Mathematics, 24.04.2020 18:08


Computers and Technology, 24.04.2020 18:08