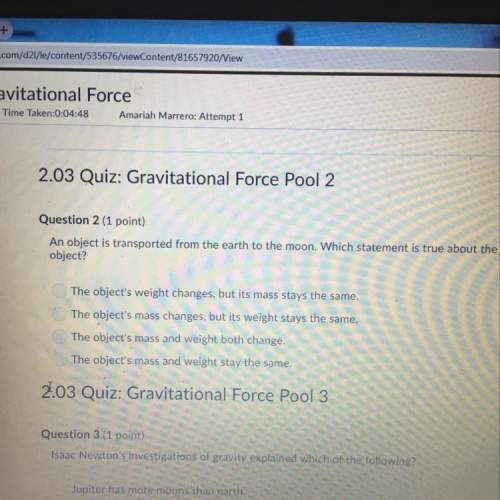
Chemistry, 14.04.2020 21:11 starpie0710
Some ionic solids become less soluble in water as the temperature is raised. If you compare the solubility of a gas in water at two different temperatures, you find the gas is more soluble at the lower temperature. The solubility of most ionic solids in water decreases as the temperature of the solution increases. The solubility of most gases in water decreases as the temperature increases because water is breaking its hydrogen bonding to the gas molecules as the temperature is raised.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3


Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1

Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1
You know the right answer?
Some ionic solids become less soluble in water as the temperature is raised. If you compare the solu...
Questions

History, 04.05.2020 23:08


Mathematics, 04.05.2020 23:08

Mathematics, 04.05.2020 23:08



English, 04.05.2020 23:08

Computers and Technology, 04.05.2020 23:08





Chemistry, 04.05.2020 23:08



Business, 04.05.2020 23:08




Mathematics, 04.05.2020 23:08