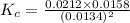Chemistry, 14.04.2020 20:01 dontworry48
Consider the following reaction: 2CH2Cl2(g) CH4(g) CCl4(g) If 0.203 moles of CH2Cl2(g), 0.323 moles of CH4, and 0.240 moles of CCl4 are at equilibrium in a 15.2 L container at 477 K, the value of the equilibrium constant, Kc, is

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3


Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1

Chemistry, 23.06.2019 06:30
Aplanet similar to earth has four moons roughly the same distance away. the moon that will most affect tides on the planet is the one that has the greatest a) mass. b) volume. c) density. d) amount of water.
Answers: 1
You know the right answer?
Consider the following reaction: 2CH2Cl2(g) CH4(g) CCl4(g) If 0.203 moles of CH2Cl2(g), 0.323 moles...
Questions


Mathematics, 16.11.2020 23:20



Health, 16.11.2020 23:20

Social Studies, 16.11.2020 23:20

Mathematics, 16.11.2020 23:20

Mathematics, 16.11.2020 23:20

History, 16.11.2020 23:20


English, 16.11.2020 23:20


Biology, 16.11.2020 23:20

History, 16.11.2020 23:20


Mathematics, 16.11.2020 23:20

Mathematics, 16.11.2020 23:20

Biology, 16.11.2020 23:20


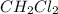 at equilibrium= 0.203 mole
at equilibrium= 0.203 mole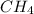 at equilibrium = 0.323 mole
at equilibrium = 0.323 mole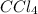 at equilibrium = 0.240mole
at equilibrium = 0.240mole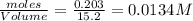
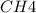 =
= 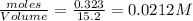
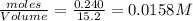
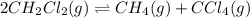
![K_c=\frac{[CH_4]\times [CCl_4]}{[CH_2Cl_2]^2}](/tpl/images/0599/0518/28fa5.png)