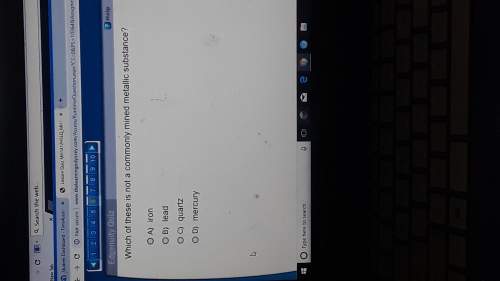
Chemistry, 14.04.2020 08:49 Jadaflournoy5
How many moles of precipitate will be formed when 50.0 mL of 0.300 M AgNO3 is reacted with excess CaI2 in the following chemical reaction? 2AgNO3(aq)+CaI2(aq) --> AgI(s)+Ca(NO3)2(aq)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 02:30
Based on the equation and the information in the table, what is the enthalpy of the reaction?
Answers: 2

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3
You know the right answer?
How many moles of precipitate will be formed when 50.0 mL of 0.300 M AgNO3 is reacted with excess Ca...
Questions





Mathematics, 27.02.2020 20:24


History, 27.02.2020 20:24

Mathematics, 27.02.2020 20:24

Chemistry, 27.02.2020 20:24

Biology, 27.02.2020 20:24



English, 27.02.2020 20:24







English, 27.02.2020 20:24