
Chemistry, 09.04.2020 00:57 ChaseRussell24
A proposed mechanism for the reaction Cl2(g) + CHCl3(g) HCl(g) + CCl4(g) in the atmosphere is Step 1: Cl2(g) 2 Cl(g) (very fast, reversible) Step 2: Cl(g) + CHCl3(g) HCl(g) + CCl3(g) (slow) Step 3: Cl(g) + CCl3(g) CCl4(g) (fast) What is the rate law for the overall reaction?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Which sentence best describes the formation of igneous rock? a- lava on the surface dries up and makes arock b_melted rocks cools and forms crystals c_rocks under tremendous heat and pressure d_magma is melted rock underground
Answers: 1

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2

Chemistry, 22.06.2019 17:00
The arrangement of particles is most ordered in a sample of
Answers: 1

Chemistry, 22.06.2019 21:00
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
A proposed mechanism for the reaction Cl2(g) + CHCl3(g) HCl(g) + CCl4(g) in the atmosphere is Step...
Questions

Spanish, 18.10.2019 19:10

English, 18.10.2019 19:10




Health, 18.10.2019 19:10












Computers and Technology, 18.10.2019 19:10


![\text{Rate}=k[Cl_2]^{1/2}[CCl_4][CHCl_3][CCl_3]^{-1}](/tpl/images/0590/9105/bc5d3.png)
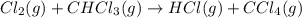
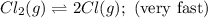
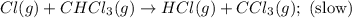
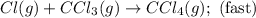
![\text{Rate}=K_2[Cl][CHCl_3]](/tpl/images/0590/9105/4079f.png) ......(1)
......(1)![K_1=\frac{[Cl]^2}{[Cl_2]}](/tpl/images/0590/9105/5c3f3.png)
![[Cl]=\sqrt{K_1[Cl_2]}](/tpl/images/0590/9105/af496.png)
![K_3=\frac{[CCl_4]}{[Cl][CCl_3]}](/tpl/images/0590/9105/b542e.png)
![[Cl]=\frac{[CCl_4]}{K_3[CCl_3]}](/tpl/images/0590/9105/a0988.png)
![\text{Rate}=K_2\times \sqrt{K_1[Cl_2]}\times \frac{[CCl_4]}{K_3[CCl_3]}\times [CHCl_3]\\\\\text{Rate}=k[Cl_2]^{1/2}[CCl_4][CHCl_3][CCl_3]^{-1}](/tpl/images/0590/9105/79491.png)


