Given the reaction represented by the balanced equation:
CH4(g)+3F2(g)->3HF(g)+CHF3 c...


Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 21:30
What is the correct name for the compound cocl3? a) cobalt(i) chloride b) cobalt(i) chlorate c) cobalt(ii) chlorate d) cobalt(iii) chloride
Answers: 1

Chemistry, 23.06.2019 01:00
Animals that reproduce sexually either do it through external or internal fertilization. read the following statement and decide if it is true or false. birds reproduce through external reproduction which is because the female will then be able to protect the egg.
Answers: 1

Chemistry, 23.06.2019 02:00
Which statement is true about the model of the electromagnetic spectrum a: it change the frequencies of light. b: it compare wavelengths of light. c: the color of light waves can be changed using the model. d: the intensities of light waves can be decreased using the model.
Answers: 2
You know the right answer?
Questions




History, 12.10.2019 12:10

Mathematics, 12.10.2019 12:10





Mathematics, 12.10.2019 12:10

English, 12.10.2019 12:10

Social Studies, 12.10.2019 12:10



Chemistry, 12.10.2019 12:10


Mathematics, 12.10.2019 12:10


English, 12.10.2019 12:10

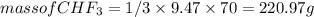
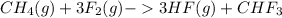
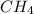 ) is taken excess amount
) is taken excess amount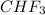 depend only on mass of fluorine
depend only on mass of fluorine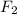 =180 g
=180 g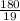 =9.47
=9.47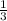 mole of
mole of 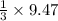 mole of
mole of 

