
Chemistry, 08.04.2020 06:40 cancerbaby209
When 1 mol of methane is burned at constant pressure, −890 kJ/mol of energy is released as heat. If a 3.72 g sample of methane is burned at constant pressure, what will be the value of ∆H? (Hint: Convert the grams of methane to moles. Also make sure your answer has the correct sign for an exothermic process.) Answer in units of kJ.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:30
The molecular formula for caffeine is cshion402. which of the following elements is not found in caffeine?
Answers: 1

Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1
You know the right answer?
When 1 mol of methane is burned at constant pressure, −890 kJ/mol of energy is released as heat. If...
Questions








Business, 02.08.2019 16:00


Business, 02.08.2019 16:00

Chemistry, 02.08.2019 16:00








Mathematics, 02.08.2019 16:00

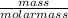
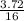 = 0.23moles
= 0.23moles 

