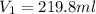Chemistry, 08.04.2020 04:33 damonsmith201615
A chemist must prepare of aqueous calcium sulfate working solution. He'll do this by pouring out some aqueous calcium sulfate stock solution into a graduated cylinder and diluting it with distilled water. Calculate the volume in of the calcium sulfate stock solution that the chemist should pour out. Round your answer to significant digits.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 03:30
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3

Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1
You know the right answer?
A chemist must prepare of aqueous calcium sulfate working solution. He'll do this by pouring out som...
Questions

Social Studies, 12.05.2021 14:50



English, 12.05.2021 14:50

History, 12.05.2021 14:50



Law, 12.05.2021 14:50




Mathematics, 12.05.2021 14:50

Mathematics, 12.05.2021 14:50


Mathematics, 12.05.2021 14:50

English, 12.05.2021 14:50

Mathematics, 12.05.2021 14:50

Mathematics, 12.05.2021 14:50

Chemistry, 12.05.2021 14:50

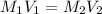
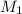 = molarity of stock calcium sulphate solution = 1.82 M
= molarity of stock calcium sulphate solution = 1.82 M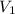 = volume of stock calcium sulphate solution = ?
= volume of stock calcium sulphate solution = ?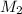 = molarity of dilute calcium sulphate solution = 1.00 M
= molarity of dilute calcium sulphate solution = 1.00 M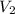 = volume of dilute calcium sulphate solution = 400 ml
= volume of dilute calcium sulphate solution = 400 ml