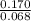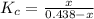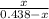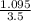Chemistry, 08.04.2020 04:41 Natavia3402
A one liter flask with the two species at equilibrium contains 0.170 mole isobutane and 0.0680 mole n-butane. Then the concentration of n-butane is increase by 0.200 moles. (0.200 mole n-butane is added.) What is the new equilibrium concentration for isobutane

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which solution is an example of a nonelectrolyte? a. iodine in hexane b. sodium nitrate in waterc. acetic acid in waterd. hydrogen chloride in water
Answers: 2

Chemistry, 22.06.2019 08:00
Match the mixture with the substance// i really need on this guys (it’s a pic btw)
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2
You know the right answer?
A one liter flask with the two species at equilibrium contains 0.170 mole isobutane and 0.0680 mole...
Questions


Mathematics, 15.12.2021 20:40



Mathematics, 15.12.2021 20:40







English, 15.12.2021 20:40



History, 15.12.2021 20:40



History, 15.12.2021 20:40


 isobutane(g)
isobutane(g)![K_{c} = \frac{[isobutane]}{[n-butane]}](/tpl/images/0588/9359/5783c.png)