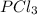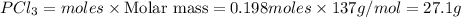For the following reaction, 6.12 grams of phosphorus (P4) are mixed with excess chlorine gas . The reaction yields 21.5 grams of phosphorus trichloride . phosphorus (P4) ( s ) chlorine ( g ) phosphorus trichloride ( l ) What is the theoretical yield of phosphorus trichloride

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 13:00
The number of neutrons is equal to the atomic number minus the atomic mass. a. true b. false
Answers: 2

Chemistry, 23.06.2019 01:00
Which statement is true regarding the diagram of circle p? the sum of y and z must be 2x. the sum of y and z must be x. the difference of z and y must be 2x. the difference of z and y must be x
Answers: 1

Chemistry, 23.06.2019 01:00
Which of the following is the molecular formula for a simple sugar? a. cooh b. h2o c. oh d. c6h12o6
Answers: 1
You know the right answer?
For the following reaction, 6.12 grams of phosphorus (P4) are mixed with excess chlorine gas . The r...
Questions

Mathematics, 08.06.2021 07:00

History, 08.06.2021 07:00


Biology, 08.06.2021 07:00

Mathematics, 08.06.2021 07:00


Geography, 08.06.2021 07:00

History, 08.06.2021 07:00








History, 08.06.2021 07:00

Mathematics, 08.06.2021 07:00

Mathematics, 08.06.2021 07:00


Physics, 08.06.2021 07:00

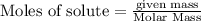
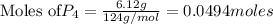
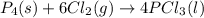
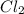 is the excess reagent,
is the excess reagent, 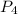 is the limiting reagent as it limits the formation of product.
is the limiting reagent as it limits the formation of product.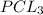
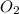 give =
give =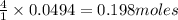 of
of