
Chemistry, 08.04.2020 01:51 habsofarah0
14. In the lab, an experimenter mixes 75.0 g of water (initially at 30oC) with 83.8 g of a solid metal (initially at 600oC). At thermal equilibrium, he measures a final temperature of 50.0oC. What metal did the experimenter probably use

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1


Chemistry, 23.06.2019 00:00
#20 which type of bond is formed when bases pair in dna? ionic bond covalent bond coordinate bond hydrogen bond
Answers: 1
You know the right answer?
14. In the lab, an experimenter mixes 75.0 g of water (initially at 30oC) with 83.8 g of a solid met...
Questions

History, 04.08.2019 19:00

Social Studies, 04.08.2019 19:00

Health, 04.08.2019 19:00

Chemistry, 04.08.2019 19:00

Spanish, 04.08.2019 19:00

Computers and Technology, 04.08.2019 19:00

Mathematics, 04.08.2019 19:00



Computers and Technology, 04.08.2019 19:00


Mathematics, 04.08.2019 19:00

Mathematics, 04.08.2019 19:00







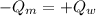
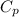

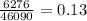 j.g⁻¹.⁰c⁻¹
j.g⁻¹.⁰c⁻¹

