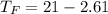Chemistry, 08.04.2020 01:15 brianna8739
Commercial cold packs consist of solid ammonium nitrate and water. NH 4NO 3( s) absorbs 25.69 kJ of heat per mole dissolved in water. In a coffee-cup calorimeter, 3.40 g NH 4NO 3( s) is dissolved in 100.0 g of water at 21.0 °C. What is the final temperature of the solution? Assume that the solution has a specific heat capacity of 4.18 J/g•K.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Explain how the integumentary system plays a crucial role in the ability to maintain homeoestasis
Answers: 1

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

Chemistry, 22.06.2019 13:30
Which of the following has wavelength longer than the wavelength of viable light? a) x rays b) gamma rays c) radios waves d) ultraviolet waves
Answers: 1

You know the right answer?
Commercial cold packs consist of solid ammonium nitrate and water. NH 4NO 3( s) absorbs 25.69 kJ of...
Questions

Mathematics, 27.04.2021 17:30

Mathematics, 27.04.2021 17:30

Mathematics, 27.04.2021 17:30

Mathematics, 27.04.2021 17:30

Geography, 27.04.2021 17:30

Mathematics, 27.04.2021 17:30


Mathematics, 27.04.2021 17:30

Mathematics, 27.04.2021 17:30





English, 27.04.2021 17:30

Mathematics, 27.04.2021 17:30

Mathematics, 27.04.2021 17:30

Mathematics, 27.04.2021 17:30


Mathematics, 27.04.2021 17:30

Biology, 27.04.2021 17:30


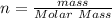

 (molar mass of Nitrogen) , 1 for
(molar mass of Nitrogen) , 1 for 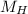 molar mass of hydrogen, 16 for
molar mass of hydrogen, 16 for 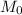 molar mass of oxygen
molar mass of oxygen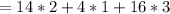
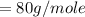
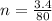


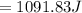

 (Specific heat capacity)
(Specific heat capacity) the subject
the subject 

 C
C