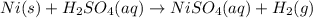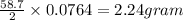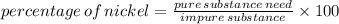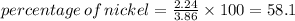Chemistry, 08.04.2020 01:38 dancemomsrule1
III. (4 points) A sample of nickel ore, which has nickel as the only metal present, is treated with an excess of sulfuric acid (H2SO4) to form nickel(II) sulfate and molecular hydrogen. (a) Write a balanced equation for the reaction. (b) If 0.0764 g of H2 is obtained from 3.86 g of the nickel ore, calculate the percent nickel, by mass, of the nickel ore.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 13:00
What is the ph of a solution with a 1.50 × 10−9 m hydroxide ion concentration?
Answers: 3


Chemistry, 22.06.2019 03:30
In saturated organic compounds, all the bonds between carbon atoms are called?
Answers: 1

You know the right answer?
III. (4 points) A sample of nickel ore, which has nickel as the only metal present, is treated with...
Questions


History, 28.08.2019 21:30




Mathematics, 28.08.2019 21:30





History, 28.08.2019 21:30


Mathematics, 28.08.2019 21:30

Mathematics, 28.08.2019 21:30


Mathematics, 28.08.2019 21:30


History, 28.08.2019 21:30


English, 28.08.2019 21:30