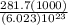Chemistry, 07.04.2020 22:28 mmassaro19
It takes 281.7 kJ of energy to remove 1 mole of electrons from the atoms on the surface of lithium metal. If lithium metal is irradiated with 261-nm light, what is the maximum kinetic energy the released electrons can have

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
An electrons position cannot be known precisely only it's probability of being in a certain location can be known
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
It takes 281.7 kJ of energy to remove 1 mole of electrons from the atoms on the surface of lithium m...
Questions

History, 18.11.2020 18:50

Computers and Technology, 18.11.2020 18:50






SAT, 18.11.2020 18:50

Physics, 18.11.2020 18:50

Geography, 18.11.2020 18:50


Physics, 18.11.2020 18:50

Biology, 18.11.2020 18:50


History, 18.11.2020 18:50

History, 18.11.2020 18:50


Mathematics, 18.11.2020 18:50


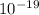 Joule
Joule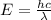 ------------ (1)
------------ (1)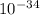 J s
J s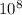
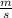
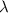 = 261 nm = 261 ×
= 261 nm = 261 × 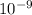 m
m