Chemistry, 07.04.2020 21:05 laura52677
G Aqueous hydrochloric acid will react with solid sodium hydroxide to produce aqueous sodium chloride and liquid water . Suppose 20. g of hydrochloric acid is mixed with 16.3 g of sodium hydroxide. Calculate the minimum mass of hydrochloric acid that could be left over by the chemical reaction. Be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:10
What approach is required to balance the objectives of sustainable development? balancing the objectives of sustainable development requires a(n) .
Answers: 3

Chemistry, 22.06.2019 09:30
Melissa is interested in her family tree and how her family has changed over its many generations. melissa probably more closely resembles
Answers: 2

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 17:30
Energy defines the different "states" of matter. in no more than 3 sentences, describe the amount of kinetic energy that each of the 3 states of matter possesses and relate that to the atom/molecular motion of each "state".
Answers: 2
You know the right answer?
G Aqueous hydrochloric acid will react with solid sodium hydroxide to produce aqueous sodium chlorid...
Questions


Mathematics, 07.01.2021 04:40

Mathematics, 07.01.2021 04:40


Mathematics, 07.01.2021 04:40

Mathematics, 07.01.2021 04:40

Mathematics, 07.01.2021 04:40

Chemistry, 07.01.2021 04:40

English, 07.01.2021 04:40


Mathematics, 07.01.2021 04:40

Mathematics, 07.01.2021 04:40

Mathematics, 07.01.2021 04:40

Chemistry, 07.01.2021 04:40


Advanced Placement (AP), 07.01.2021 04:40

Physics, 07.01.2021 04:40



Mathematics, 07.01.2021 04:40

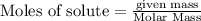
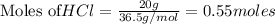
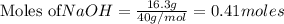
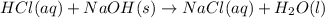
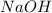 require = 1 mole of
require = 1 mole of 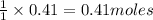 of
of 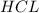 is the excess reagent.
is the excess reagent.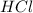 left =
left =