
Chemistry, 07.04.2020 20:09 leelee85503
A 20.00 ml sample of calcium hydroxide is titrated with 0.0100 M HCl until the color of the phenolphthalein indicator just disappears. The following readings were made on the buret: Initial reading = 2.77 ml Final reading = 30.23 ml What is the calculated Ksp of the calcium hydroxide?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3


Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2

Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1
You know the right answer?
A 20.00 ml sample of calcium hydroxide is titrated with 0.0100 M HCl until the color of the phenolph...
Questions

Mathematics, 29.11.2020 04:10


Social Studies, 29.11.2020 04:10

Advanced Placement (AP), 29.11.2020 04:10


Mathematics, 29.11.2020 04:10


English, 29.11.2020 04:10

English, 29.11.2020 04:20

English, 29.11.2020 04:20

Mathematics, 29.11.2020 04:20

Mathematics, 29.11.2020 04:20

Spanish, 29.11.2020 04:20


English, 29.11.2020 04:20

English, 29.11.2020 04:20


Social Studies, 29.11.2020 04:20

Mathematics, 29.11.2020 04:20

Arts, 29.11.2020 04:20

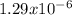
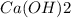 so:
so: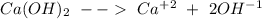 Reaction 1
Reaction 1![Ksp~=~[Ca^+^2][OH^-^1]^2](/tpl/images/0587/1033/57543.png)
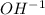 and
and 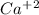 concentrations. These concentrations can be calculate using the titration procedure. So, we have to start with the ionic reaction between HCl and Ca(OH)2, so:
concentrations. These concentrations can be calculate using the titration procedure. So, we have to start with the ionic reaction between HCl and Ca(OH)2, so: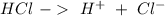 Reaction 2
Reaction 2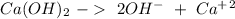 Reaction 3
Reaction 3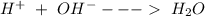 Reaction4
Reaction4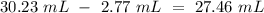
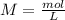
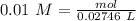
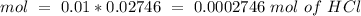
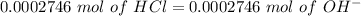
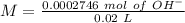
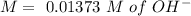
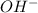 (reaction 1), the molar ratio is 1:2 therefore:
(reaction 1), the molar ratio is 1:2 therefore: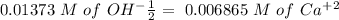
![Ksp=~[0.006865][0.01373]^2~=~1.29x10^-^6](/tpl/images/0587/1033/85932.png)
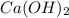 is
is 

