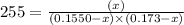For the following reaction, KcKc = 255 at 1000 KK.
CO (g) + Cl2 (g) ⇌ COCl2 (g)CO (g) + Cl2 (g) ⇌ COCl2 (g)
A reaction mixture initially contains a COCO concentration of 0.1550 MM and a Cl2Cl2 concentration of 0.173 MM at 1000 KK.
a) What is the equilibrium concentration of COCO at 1000 KK?
b) What is the equilibrium concentration of Cl2Cl2 at 1000 KK?
c) What is the equilibrium concentration of COCl2COCl2 at 1000 KK?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:20
Asolution contains 180 g of glucose (c6h12o6) and 162 g of water. what is the mole fraction of glucose?
Answers: 3

Chemistry, 22.06.2019 12:30
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3

Chemistry, 23.06.2019 01:00
Which statement is true regarding the diagram of circle p? the sum of y and z must be 2x. the sum of y and z must be x. the difference of z and y must be 2x. the difference of z and y must be x
Answers: 1

Chemistry, 23.06.2019 09:00
Need ! assume that the variables x and y are directly related. if k = 8, what is the value for each of the following points? be sure and record your data to be used in the following problem. x y k 0.
Answers: 2
You know the right answer?
For the following reaction, KcKc = 255 at 1000 KK.
CO (g) + Cl2 (g) ⇌ COCl2 (g)CO (g) + Cl2 (g...
CO (g) + Cl2 (g) ⇌ COCl2 (g)CO (g) + Cl2 (g...
Questions

Physics, 01.02.2021 06:50


History, 01.02.2021 06:50




Spanish, 01.02.2021 06:50


Mathematics, 01.02.2021 06:50

Mathematics, 01.02.2021 06:50

English, 01.02.2021 06:50









Mathematics, 01.02.2021 06:50

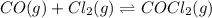

![K_c=\frac{[COCl_2]}{[CO][Cl_2]}](/tpl/images/0587/0731/36d91.png)