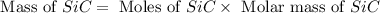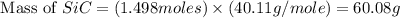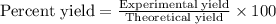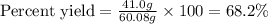Equation: SiO2 + 3C = SiC + 2CO When 90.0 g of silicon dioxide is heated with an excess of carbon, 41.0 g of silicon carbide is produced. What is the percent yield of this reaction? (find the theoretical amount of SiC using stoichiometry, then calculate percent yield)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 23.06.2019 00:00
What is the pressure of 0.500 moles of carbon dioxide gas in a 2.5 l tank and at a temperature of 301 k? (r=0.0821 l·atm/mol·k) 3.08 atm 1.2 atm 0.23 atm 4.01 atm 4.94 atm
Answers: 1

Chemistry, 23.06.2019 00:30
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1

Chemistry, 23.06.2019 00:30
An ice cube with a volume of 45.0ml and a density of 0.9000g/cm3 floats in a liquid with a density of 1.36g/ml. what volume of the cube is submerged in the liquid
Answers: 3
You know the right answer?
Equation: SiO2 + 3C = SiC + 2CO When 90.0 g of silicon dioxide is heated with an excess of carbon, 4...
Questions

History, 19.07.2019 13:40


Mathematics, 19.07.2019 13:40

Geography, 19.07.2019 13:40






Mathematics, 19.07.2019 13:40

Mathematics, 19.07.2019 13:40




History, 19.07.2019 13:40



Social Studies, 19.07.2019 13:40


Mathematics, 19.07.2019 13:40

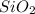 = 90.0 g
= 90.0 g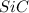 = 41.0 g
= 41.0 g
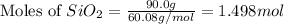
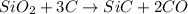
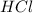 react to give 1.498 mole of
react to give 1.498 mole of