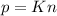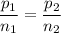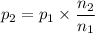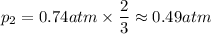A gas exerts a pressure of 0.74 atm in a certain container. Suddenly, a chemical change occurs that consumes half of the molecules originally present and forms two new molecules for every three consumed. Determine the new pressure in the container if the volume of the container and the temperature are unchanged. The reaction that occurs is: 3A(g) —> 2B(g)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 22.06.2019 22:00
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1

Chemistry, 23.06.2019 01:00
If a sample of radioactive isotopes takes 600 minutes to decay from 400 grams to 50 grams, what is the half-life of the isotope?
Answers: 1

You know the right answer?
A gas exerts a pressure of 0.74 atm in a certain container. Suddenly, a chemical change occurs that...
Questions

Mathematics, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

English, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

History, 13.09.2020 23:01

Physics, 13.09.2020 23:01

Social Studies, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

Health, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01

Chemistry, 13.09.2020 23:01

Health, 13.09.2020 23:01

Chemistry, 13.09.2020 23:01

History, 13.09.2020 23:01

Mathematics, 13.09.2020 23:01