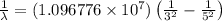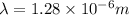Line spectra from all regions of the electromagnetic spectrum, including the Paschen series of infrared lines for hydrogen, are used by astronomers to identify elements present in the atmospheres of stars. Calculate the wavelength of the photon emitted when the hydrogen atom undergoes a transition from n = 5 to n = 3. (R = 1.096776 × 107 m–1)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 13:00
Asolution has a ca2+ concentration of 0.049 m and an f- concentration is 0.147 m at equilibrium. the value of ksp for caf2 at 25°c is 4.0 x 10-11. will this solution form a precipitate? yes no
Answers: 3


Chemistry, 22.06.2019 11:00
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2

Chemistry, 22.06.2019 12:00
Hey guys so i need to know what is _nh3+> nh4oh ~chemistry~
Answers: 1
You know the right answer?
Line spectra from all regions of the electromagnetic spectrum, including the Paschen series of infra...
Questions

Chemistry, 27.10.2019 18:43






Mathematics, 27.10.2019 18:43



Mathematics, 27.10.2019 18:43

History, 27.10.2019 18:43





History, 27.10.2019 18:43




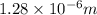
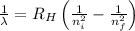
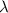 = Wavelength of radiation
= Wavelength of radiation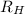 = Rydberg's Constant = 1.096776 × 10⁷ m⁻¹
= Rydberg's Constant = 1.096776 × 10⁷ m⁻¹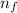 = Higher energy level = 5
= Higher energy level = 5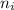 = Lower energy level = 3
= Lower energy level = 3