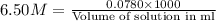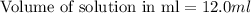Chemistry, 07.04.2020 15:52 boweytom6217
Zinc reacts with hydrochloric acid according to the reaction equation Zn ( s ) + 2 HCl ( aq ) ⟶ ZnCl 2 ( aq ) + H 2 ( g ) How many milliliters of 6.50 M HCl ( aq ) are required to react with 2.55 g Zn ( s ) ?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 23.06.2019 04:31
2ki + pb(no3)2 → 2kno3 + pbi2 determine how many moles of kno3 are created if 0.03 moles of ki are completely consumed.
Answers: 1

You know the right answer?
Zinc reacts with hydrochloric acid according to the reaction equation Zn ( s ) + 2 HCl ( aq ) ⟶ ZnCl...
Questions


Chemistry, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01


Mathematics, 06.10.2020 14:01


Mathematics, 06.10.2020 14:01

English, 06.10.2020 14:01


Biology, 06.10.2020 14:01




Mathematics, 06.10.2020 14:01

English, 06.10.2020 14:01

History, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01


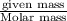
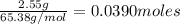
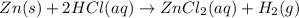
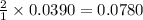 moles of HCl
moles of HCl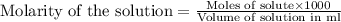 .....(1)
.....(1)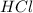 solution = 6.50 M
solution = 6.50 M