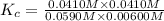Chemistry, 07.04.2020 03:15 sethlynn2003
Calculate the equilibrium constant for the reaction using the balanced chemical equation and the concentrations of the substances at equilibrium.
Use the
appropriate significant figures in reporting the answers.
CO(g) + H2O(g) ⇌ CO2(g) + H2(g) [CO] = 0.0590 M; [H2O] = 0.00600 M;
[CO2] = 0.0410 M; [H2] = 0.0410 M
K =-

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Agood hypothesis includes which of the following? a: prediction b: data c: uncertainty d: conclusion
Answers: 1



Chemistry, 22.06.2019 05:00
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1
You know the right answer?
Calculate the equilibrium constant for the reaction using the balanced chemical equation and the con...
Questions

Biology, 09.03.2020 08:21

Mathematics, 09.03.2020 08:21


History, 09.03.2020 08:24

Mathematics, 09.03.2020 08:27


Health, 09.03.2020 08:28

Mathematics, 09.03.2020 08:28

Mathematics, 09.03.2020 08:28


Mathematics, 09.03.2020 08:31

Physics, 09.03.2020 08:31


Law, 09.03.2020 08:33

Mathematics, 09.03.2020 08:34


Mathematics, 09.03.2020 08:34


Mathematics, 09.03.2020 08:36

History, 09.03.2020 08:37

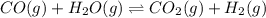
![[CO]=0.0590 M,[H_2O]=0.00600 M](/tpl/images/0585/8718/63397.png)
![[CO_2]=0.0410 M,[H_2]=0.0410 M](/tpl/images/0585/8718/efcda.png)
![K_c=\frac{[CO_2][H_2]}{[CO][H_2O]}](/tpl/images/0585/8718/fbbde.png)