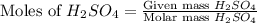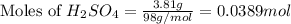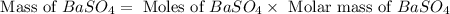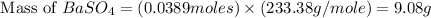Chemistry, 07.04.2020 01:26 eaalvarezelisa01
For the following reaction, 3.81 grams of sulfuric acid are mixed with excess barium hydroxide. The reaction yields 8.45 grams of barium sulfate.
barium hydroxide (aq) + sulfuric acid (aq) ---> barium sulfate (s) + water (l)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘c to water at 25.0 ∘c? show work and constants, trying to figure out how it works. only given the heat capacity for steam and water so try to only use that
Answers: 1


Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1

Chemistry, 22.06.2019 18:00
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1
You know the right answer?
For the following reaction, 3.81 grams of sulfuric acid are mixed with excess barium hydroxide. The...
Questions

Mathematics, 25.04.2022 14:00

Mathematics, 25.04.2022 14:00


Mathematics, 25.04.2022 14:00


Mathematics, 25.04.2022 14:00



Mathematics, 25.04.2022 14:00






Computers and Technology, 25.04.2022 14:00



SAT, 25.04.2022 14:00

Mathematics, 25.04.2022 14:00

Computers and Technology, 25.04.2022 14:20

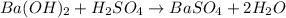
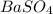 is, 9.08 grams.
is, 9.08 grams.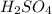 = 3.81 g
= 3.81 g