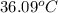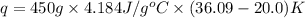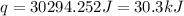150.0 mL of 2.00 M Pb(NO3)2 solution is mixed with 300.0 mL of 2.00 M NaI solution in a coffee cup calorimeter of negligible heat capacity.
The initial temperature of the two solutions are both at 20.00 oC, the final temperature of the mixed solution is 36.09 oC.
c of H 2O = 4.184 J/g oC ; d of solution is 1.00g/mL. Refer to the following:
Pb(NO3)2(aq) + 2NaI (aq) --> PbI2(s) + 2NaNO3(aq)
What is q of the water? Please watch units. Answers have units of kJ. You have to convert J to kJ.
-1790 kJ/mol
42.9 kJ/mol
- 451 kJ/mol
30.3 kJ/mol
20.9 kJ/mol
91.7 kJ/mol
85.4 kJ/mol

Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 21:50
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1
You know the right answer?
150.0 mL of 2.00 M Pb(NO3)2 solution is mixed with 300.0 mL of 2.00 M NaI solution in a coffee cup c...
Questions







Social Studies, 22.05.2020 00:06





Mathematics, 22.05.2020 00:06


History, 22.05.2020 00:06

Mathematics, 22.05.2020 00:06



History, 22.05.2020 00:06

English, 22.05.2020 00:06

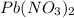 and NaI.
and NaI.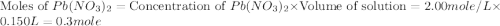
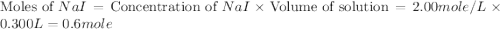
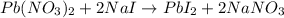
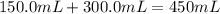
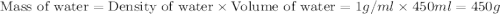
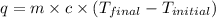
 = specific heat of water =
= specific heat of water = 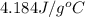
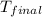 = final temperature of water =
= final temperature of water = 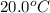
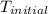 = initial temperature of metal =
= initial temperature of metal =