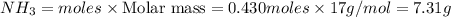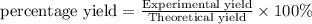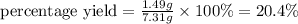Chemistry, 06.04.2020 18:26 gunner20115
The Haber-Bosch process is a very important industrial process. In the Haber-Bosch process, hydrogen gas reacts with nitrogen gas to produce ammonia according to the equation 3H2(g)+N2(g)→2NH3(g)3H2(g)+N2(g)→2N H3(g) The ammonia produced in the Haber-Bosch process has a wide range of uses, from fertilizer to pharmaceuticals. However, the production of ammonia is difficult, resulting in lower yields than those predicted from the chemical equation.1.29g H2 is allowed to react with 9.55g N2, producing 1.49g NH3.
What is the theoretical yield for this reaction under the given conditions?
What is the percent yield for this reaction under the given conditions?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 05:30
The climate of the continental united states is generally 1. tropical 2. temperate 3. arctic 4. highland
Answers: 1


Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1
You know the right answer?
The Haber-Bosch process is a very important industrial process. In the Haber-Bosch process, hydrogen...
Questions




Advanced Placement (AP), 24.02.2022 15:30

Mathematics, 24.02.2022 15:30



Health, 24.02.2022 15:30

Physics, 24.02.2022 15:30

English, 24.02.2022 15:30


Mathematics, 24.02.2022 15:40

Computers and Technology, 24.02.2022 15:40




Biology, 24.02.2022 15:40



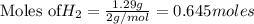
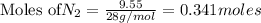
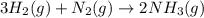
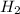 require = 1 mole of
require = 1 mole of 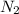
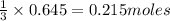 of
of 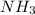
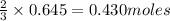 of
of