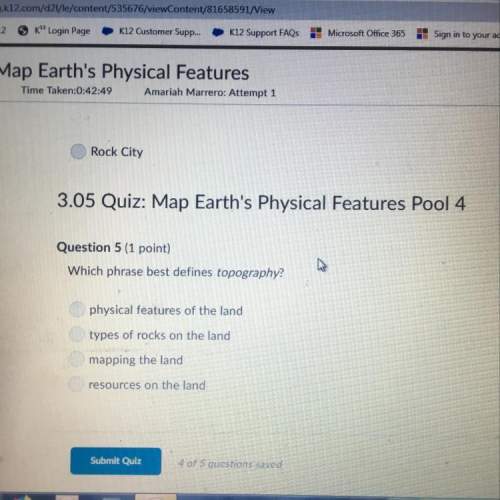
Chemistry, 06.04.2020 00:57 putaprincess16
The combustion of methane(CH4) produces carbon dioxide and water. Assume that 2.0 mol of CH4 burned in the presence of excess air. What is the percentage yield if in an experiment the reaction produces 87.0 g of CO2?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1


You know the right answer?
The combustion of methane(CH4) produces carbon dioxide and water. Assume that 2.0 mol of CH4 burned...
Questions


Mathematics, 08.10.2021 14:00



Mathematics, 08.10.2021 14:00

Biology, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00


Mathematics, 08.10.2021 14:00


English, 08.10.2021 14:00


English, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00



Chemistry, 08.10.2021 14:00


Mathematics, 08.10.2021 14:00