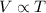Chemistry, 04.04.2020 18:25 ravenjade2395
Jorge inflates a beach ball to a volume of 4L in his air-conditioned house where the temperature is 18℃. That afternoon he takes the beach ball to the beach with some friends. The temperature at the beach is 32℃, and the air pressure at the beach is the same as it was at Jorge's house. What will happen to Jorge's beach ball when he is at the beach? Choose the claim that best answers the question: * 1 point The beach ball will get smaller at the beach because the molecules are moving slower. The beach ball will stay the same size at the beach because the pressure is constant. The beach ball will get larger at the beach because the molecules are moving faster. The beach ball will get larger at the beach because the pressure will cause it to expand. The beach ball will get smaller at the beach because the molecules will collide less often.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2
You know the right answer?
Jorge inflates a beach ball to a volume of 4L in his air-conditioned house where the temperature is...
Questions



Mathematics, 24.08.2020 02:01














Mathematics, 24.08.2020 02:01



Mathematics, 24.08.2020 02:01