Chemistry, 04.04.2020 14:34 wirchakethan23
The reform reaction between steam and gaseous methane () produces "synthesis gas," a mixture of carbon monoxide gas and dihydrogen gas. Synthesis gas is one of the most widely used industrial chemicals, and is the major industrial source of hydrogen. Suppose a chemical engineer studying a new catalyst for the reform reaction finds that liters per second of methane are consumed when the reaction is run at and . Calculate the rate at which dihydrogen is being produced. Give your answer in kilograms per second. Round your answer to significant digits..

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 06:30
The best solution for preventing harm to people and pets from severe hurricanes involves determining and warning residents about what
Answers: 1


Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2
You know the right answer?
The reform reaction between steam and gaseous methane () produces "synthesis gas," a mixture of carb...
Questions

Physics, 20.10.2020 01:01


Mathematics, 20.10.2020 01:01



Computers and Technology, 20.10.2020 01:01

Biology, 20.10.2020 01:01


Mathematics, 20.10.2020 01:01

Computers and Technology, 20.10.2020 01:01


Mathematics, 20.10.2020 01:01

Mathematics, 20.10.2020 01:01


Biology, 20.10.2020 01:01

Biology, 20.10.2020 01:01


Mathematics, 20.10.2020 01:01

History, 20.10.2020 01:01

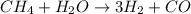
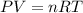
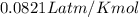
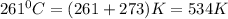
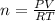
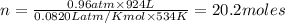
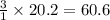 moles of hydrogen
moles of hydrogen