
Chemistry, 04.04.2020 14:28 shaydog6353
Consider the reaction between 15.0 mL of a 1.00 M aqueous solution of AgNO3 and 10.0 mL of a 1.00 M aqueous solution of K2CrO4. When these react, a precipitate is observed. What is present in solution after the reaction is complete

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

You know the right answer?
Consider the reaction between 15.0 mL of a 1.00 M aqueous solution of AgNO3 and 10.0 mL of a 1.00 M...
Questions




English, 06.11.2020 16:20










Computers and Technology, 06.11.2020 16:20






Mathematics, 06.11.2020 16:20

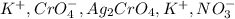
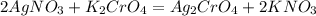
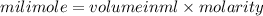
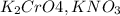 are soluble in water so it will dissociate in the solution
are soluble in water so it will dissociate in the solution

