
Chemistry, 04.04.2020 13:17 mahoganyking16
Calculate the [H+] in a solution that is 0.803 M in NaX and 0.677 M in HX given that the Ka of HX is 8.64 ⋅ 10 − 7 8.64⋅10-7. Report your answer in scientific notation to 3 sig figs.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Write the overall equation for the reaction for lithium battery
Answers: 2

Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 22.06.2019 19:40
What causes different colors to appear in the sky? the absorption of light by air molecules the reflection of light by bodies of water the greenhouse effect in earth's atmosphere the scattering and reflection of light by dust particles
Answers: 2
You know the right answer?
Calculate the [H+] in a solution that is 0.803 M in NaX and 0.677 M in HX given that the Ka of HX is...
Questions



Mathematics, 05.11.2020 07:40



Mathematics, 05.11.2020 07:40



Biology, 05.11.2020 07:40


Mathematics, 05.11.2020 07:40

Mathematics, 05.11.2020 07:40


History, 05.11.2020 07:40


English, 05.11.2020 07:40

Chemistry, 05.11.2020 07:40

Mathematics, 05.11.2020 07:40

Mathematics, 05.11.2020 07:40

English, 05.11.2020 07:40

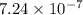 .
.![pH=pK_a+\log(\frac{[salt]}{[acid]})](/tpl/images/0582/4215/e4eea.png)
![pH=pK_a+\log(\frac{[NaX]}{[HX]})](/tpl/images/0582/4215/da324.png)
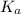 = Acid dissociation constant of HX =
= Acid dissociation constant of HX = 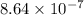
![pK_a=-\log[K_a]=-\log[8.64\times 10^{-7}]=6.06](/tpl/images/0582/4215/ec5c6.png)
![[salt]=[NaX]=0.803 M](/tpl/images/0582/4215/da1f0.png)
![[acid]=[HX]=0.677 M](/tpl/images/0582/4215/836f0.png)
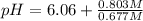
![pH=-\log[H^+]](/tpl/images/0582/4215/cf945.png)
![6.14=-\log[H^+]](/tpl/images/0582/4215/0a3f8.png)
![[H^+]=10^{-6.14}=7.24\times 10^{-7}](/tpl/images/0582/4215/e51b5.png)


