Chemistry, 04.04.2020 11:36 kiragor2545
The percent by mass of bicarbonate (HCO3−) in a certain Alka-Seltzer product is 32.5 percent. Calculate the volume of CO2 generated (in mL) at 37°C and 1.00 atm if a person were to accidentally ingest a 3.45-g tablet without following instructions. (Hint: The reaction occurs between HCO3− and HCl acid in the stomach.)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:30
If a 60-g object has a volume of 30 cm3, what is its density? 2 g/cm3 0.5 cm3/g 1800 g * cm3 none of the above
Answers: 3

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 23.06.2019 00:00
In an exothermic reaction, energy may be released to the surroundings in the form of question 4 options: heat light thermal all of the above
Answers: 3

Chemistry, 23.06.2019 02:00
Butane gas reacts with oxygen gas to give carbon dioxide gas and water vapor (gas). if you mix butane and oxygen in the correct stoichiometric ratio, and if the total pressure of the mixture is 390 mmhg, what is the pressure (in mmhg) of water vapor after the reaction is completed (temperature and volume do not change).
Answers: 2
You know the right answer?
The percent by mass of bicarbonate (HCO3−) in a certain Alka-Seltzer product is 32.5 percent. Calcul...
Questions

Biology, 29.08.2021 05:00




Mathematics, 29.08.2021 05:00

Mathematics, 29.08.2021 05:00

Health, 29.08.2021 05:00


Mathematics, 29.08.2021 05:00


Mathematics, 29.08.2021 05:00

Mathematics, 29.08.2021 05:00


Mathematics, 29.08.2021 05:00

Health, 29.08.2021 05:00


Mathematics, 29.08.2021 05:00

Computers and Technology, 29.08.2021 05:00


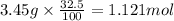
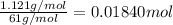
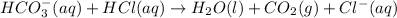
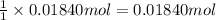 of carbon dioxide gas
of carbon dioxide gas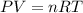 (ideal gas equation)
(ideal gas equation)