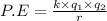Assuming that the distances between the two ions are the same in all cases, which of the following ion pairs has the greatest electrostatic potential energy (i. e., largest in magnitude)? Please explain your answer. a.) Na+ - Cl- b.) Na+ - O-2. c.) Al+3 - O-2. d.) Mg+2-O-2 e.) Na- -Mg+2

Answers: 3
Another question on Chemistry


Chemistry, 23.06.2019 02:30
Ascientist wants to know how individual lions within a pride interact with each other in their own environment. to do this, the scientist sedates and tags all of the lions within a pride. then, he places several remotely-controlled video cameras near the lions' den and performs an observational field study. he collects continuous video footage over the span of one year, analyzes the video, and then forms conclusions based on his observations.
Answers: 2


Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2
You know the right answer?
Assuming that the distances between the two ions are the same in all cases, which of the following i...
Questions


Mathematics, 10.12.2020 18:30

Advanced Placement (AP), 10.12.2020 18:30


Health, 10.12.2020 18:30

Mathematics, 10.12.2020 18:30

Mathematics, 10.12.2020 18:30

History, 10.12.2020 18:30

History, 10.12.2020 18:30

Mathematics, 10.12.2020 18:30

Arts, 10.12.2020 18:30



Health, 10.12.2020 18:30


Biology, 10.12.2020 18:30


Mathematics, 10.12.2020 18:30