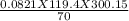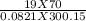A 70-liter tank of oxygen gas at 27 degree Celcius and 42.0 atm springs a leak overnight. when the tank was found in the morning, the pressure in the tank had dropped to 19.0 atm. If the tank originally held 119.4 moles of oxygen gas, how many moles of gas were left the next morning, assuming the temperature and volume of the rank stayed constant?.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 22:00
If a solution contains 3 moles/liter of sodium chloride (nacl, made of sodium ions and chloride ions), what is the osmolarity of this solution
Answers: 3


Chemistry, 23.06.2019 16:20
Which of the following subject areas contains questions that can be answered by science? alchemy ethics forensics politics
Answers: 1

Chemistry, 23.06.2019 19:30
Is the following chemical equation balanced? agno3 + nacl 4agcl + nano3 yes no
Answers: 1
You know the right answer?
A 70-liter tank of oxygen gas at 27 degree Celcius and 42.0 atm springs a leak overnight. when the t...
Questions

Mathematics, 01.03.2021 04:40



Mathematics, 01.03.2021 04:40



Mathematics, 01.03.2021 04:40


Chemistry, 01.03.2021 04:40

Mathematics, 01.03.2021 04:40



History, 01.03.2021 04:40


Mathematics, 01.03.2021 04:40

Mathematics, 01.03.2021 04:40


Mathematics, 01.03.2021 04:40


Mathematics, 01.03.2021 04:40