
Chemistry, 04.04.2020 11:01 deebroussard
Find the pH during the titration of 20.00 mL of 0.1000 M butanoic acid, CH3CH2CH2COOH (Ka = 1.54 x 10^-5), with 0.1000 M NaOH solution after the following additions of titrant.
(a) 10.00 mL: pH =
(b) 20.10 mL: pH =
(c) 25.00 mL: pH =

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Look at the bean data from days 4–6. use these data to explain how natural selection changed the number of dark red walking beans over time. writing part
Answers: 3

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3
You know the right answer?
Find the pH during the titration of 20.00 mL of 0.1000 M butanoic acid, CH3CH2CH2COOH (Ka = 1.54 x 1...
Questions

Mathematics, 21.08.2019 23:30


Social Studies, 21.08.2019 23:30

History, 21.08.2019 23:30






Mathematics, 21.08.2019 23:30

Physics, 21.08.2019 23:30

Mathematics, 21.08.2019 23:30

Biology, 21.08.2019 23:30

Business, 21.08.2019 23:30

Social Studies, 21.08.2019 23:30

Health, 21.08.2019 23:30

English, 21.08.2019 23:30


Mathematics, 21.08.2019 23:30

Physics, 21.08.2019 23:30

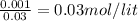
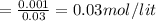
![pH=pK_{a}+log\frac{[conjugate base]}{[acid]} \\\\=-log(1.54X10^{-5} )+log\frac{0.03}{0.03} \\\\=4.81](/tpl/images/0582/2071/3377e.png)




