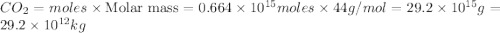Assume that all of this fossil fuel is in the form of octane (C8H18) and calculate how much CO2 in kilograms is produced by world fossil fuel combustion per year. (Hint: Begin by writing a balanced equation for the combustion of octane.) Express your answer using two significant figures.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2



You know the right answer?
Assume that all of this fossil fuel is in the form of octane (C8H18) and calculate how much CO2 in k...
Questions

Biology, 05.11.2019 23:31

History, 05.11.2019 23:31


History, 05.11.2019 23:31

Mathematics, 05.11.2019 23:31


Social Studies, 05.11.2019 23:31













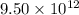 kg of petroleum per year. Assume that all of this petroleum is in the form of octane. Calculate how much CO2 in kilograms is produced by world fossil fuel combustion per year.( Hint: Begin by writing a balanced equation for the combustion of octane.)
kg of petroleum per year. Assume that all of this petroleum is in the form of octane. Calculate how much CO2 in kilograms is produced by world fossil fuel combustion per year.( Hint: Begin by writing a balanced equation for the combustion of octane.)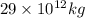
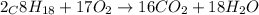
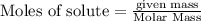
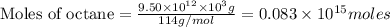
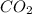
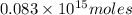 of octane give =
of octane give =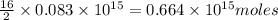 of
of