Chemistry, 04.04.2020 10:46 clonetrooper099
The molar enthalpy of vaporization of water is 40.79 kJ/mol, and the molar enthalpy of fusion of ice is 6.009 kJ/mol. The molar mass of water is 18.02 g/mol. How much energy is absorbed when 30.3 g of liquid water boils

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2


Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 22.06.2019 19:30
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1
You know the right answer?
The molar enthalpy of vaporization of water is 40.79 kJ/mol, and the molar enthalpy of fusion of ice...
Questions


English, 24.12.2020 14:00



Mathematics, 24.12.2020 14:00


Mathematics, 24.12.2020 14:00


Mathematics, 24.12.2020 14:00



Mathematics, 24.12.2020 14:00

Health, 24.12.2020 14:00



Computers and Technology, 24.12.2020 14:00

English, 24.12.2020 14:00



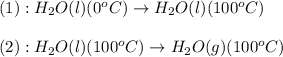
![Q=[m\times c_{p,l}\times (T_{final}-T_{initial})]+[m\times \Delta H_{vap}]](/tpl/images/0582/1538/e3ba2.png)
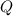 = heat required for the reaction = ?
= heat required for the reaction = ?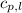 = specific heat of liquid water =
= specific heat of liquid water = 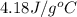
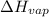 = enthalpy change for vaporization =
= enthalpy change for vaporization = 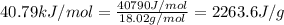
![Q=[30.3g\times 4.18J/g^oC\times (100-0)^oC]+[30.3g\times 2263.6J/g]](/tpl/images/0582/1538/57d8f.png)