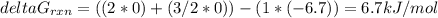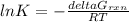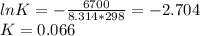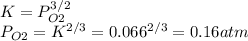Chemistry, 04.04.2020 09:43 emmarieasimon
Consider the decomposition of a metal oxide to its elements, where M represents a generic metal. M 3 O 4 ( s ) − ⇀ ↽ − 3 M ( s ) + 2 O 2 ( g ) What is the standard change in Gibbs energy for the reaction, as written, in the forward direction? Δ G ∘ rxn = kJ/mol What is the equilibrium constant of this reaction, as written, in the forward direction at 298 K? K = What is the equilibrium pressure of O2(g) over M(s) at 298 K? P O 2 = atm

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Agas can holds 2.0 gal of gasoline. what is this quantity in cubic centimeters?
Answers: 2

Chemistry, 23.06.2019 03:30
In general metals get as you move from left to right across the periodic table.
Answers: 1

Chemistry, 23.06.2019 06:30
Which of the following is true about the products formed during photosynthesis? (5 points) select one: a. they have the same mass as the mass of reactants. b. they are the same set of compounds as the reactants. c. they have more mass than the mass of reactants. d. they are chemically the same as the reactants.
Answers: 1

Chemistry, 23.06.2019 07:30
Which of the following statements best explains why chemistry is testable a) it can measure data by experiments b) it cannot add new evidence c) it cannot be verified d) it is biased
Answers: 1
You know the right answer?
Consider the decomposition of a metal oxide to its elements, where M represents a generic metal. M 3...
Questions


Mathematics, 06.05.2021 05:20



Mathematics, 06.05.2021 05:20

Computers and Technology, 06.05.2021 05:20


Mathematics, 06.05.2021 05:20

Mathematics, 06.05.2021 05:20



Mathematics, 06.05.2021 05:20

Mathematics, 06.05.2021 05:20

Mathematics, 06.05.2021 05:20

Mathematics, 06.05.2021 05:20



Mathematics, 06.05.2021 05:20

Mathematics, 06.05.2021 05:20

Mathematics, 06.05.2021 05:20