
Chemistry, 04.04.2020 10:06 bluebunny1231999
Calculate the pH for the following weak acid. A solution of HCOOH has 0.12M HCOOH at equilibrium. The Ka for HCOOH is 1.8×10−4. What is the pH of this solution at equilibrium?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural.question 2 reflects a moral or social value.question 3 refers to something that can be measured.question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1
You know the right answer?
Calculate the pH for the following weak acid. A solution of HCOOH has 0.12M HCOOH at equilibrium. Th...
Questions



History, 03.08.2019 11:30

History, 03.08.2019 11:30

History, 03.08.2019 11:30

History, 03.08.2019 11:30

History, 03.08.2019 11:30

History, 03.08.2019 11:30


Chemistry, 03.08.2019 11:30



Social Studies, 03.08.2019 11:30



Geography, 03.08.2019 11:40

Spanish, 03.08.2019 11:40

Social Studies, 03.08.2019 11:40


English, 03.08.2019 11:40

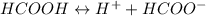
![\Ka= \frac{[H^+][HCOO^-]}{[HCOOH]}](/tpl/images/0582/0861/d7b9e.png)
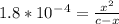
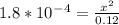
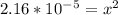
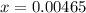
![[H^+] .](/tpl/images/0582/0861/74f2d.png)
![[H^+] = 0.00465 M](/tpl/images/0582/0861/817d9.png)
![pH=-log[H^+]](/tpl/images/0582/0861/15713.png)
![K_{a} = \frac{[H^{+} ][HCOO^{-} ]}{[HCOOH]}](/tpl/images/0582/0861/8c4ca.png)
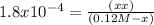
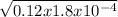 = 4.65 x 10⁻³
= 4.65 x 10⁻³

