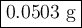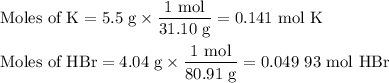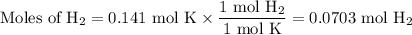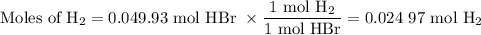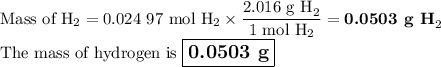2K + 2HBr → 2 KBr + H2
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hyd...

Chemistry, 03.04.2020 06:24 spdesch2558
2K + 2HBr → 2 KBr + H2
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hydrogen gas(H₂)
●a). What is the limiting reactant?
●b.)What is the excess reactant?
●C.)How much product is produced?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3


Chemistry, 23.06.2019 07:30
Using this reversible reaction, answer the questions below: n2o4 2no2 (colorless) (reddish-brown) -as the temperature increased, what happened to the n2o4 concentration? -was the formation of reactants or products favored by the addition of heat? -which reaction is exothermic? right to left or left to right? -if the change of enthalpy of this reaction when proceeding left to right is 14 kcal, which chemical equation is correct? n2o4 2no2 + 14 kcal n2o4 2no2, hr = +14 kcal n2o4 + 14 kcal 2no2 n2o4 2no2, hr = -14 kcal
Answers: 1
You know the right answer?
Questions





Mathematics, 09.03.2021 16:40

Mathematics, 09.03.2021 16:40

Mathematics, 09.03.2021 16:40

Mathematics, 09.03.2021 16:40



Mathematics, 09.03.2021 16:40

English, 09.03.2021 16:40

Mathematics, 09.03.2021 16:40


Biology, 09.03.2021 16:40

Chemistry, 09.03.2021 16:40


Mathematics, 09.03.2021 16:40


Mathematics, 09.03.2021 16:40