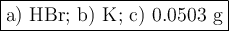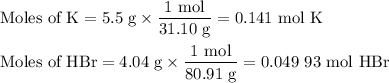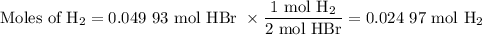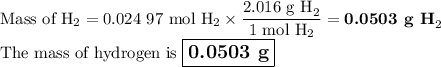2K + 2HBr → 2 KBr + H2
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hydrogen gas(H₂)
●a). What is the limiting reactant?
●b.)What is the excess reactant?
2K + 2HBr → 2 KBr + H2
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hydrogen gas(H₂)
●a). What is the limiting reactant?
●b.)What is the excess reactant?
2K + 2HBr → 2 KBr + H2
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hydrogen gas(H₂)
●a). What is the limiting reactant?
●b.)What is the excess reactant?
●C.)How much product is produced?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2

Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3
You know the right answer?
2K + 2HBr → 2 KBr + H2
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hyd...
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hyd...
Questions


Business, 09.11.2021 21:10

Mathematics, 09.11.2021 21:10



Advanced Placement (AP), 09.11.2021 21:10

Physics, 09.11.2021 21:10

History, 09.11.2021 21:10

English, 09.11.2021 21:10

Physics, 09.11.2021 21:10

English, 09.11.2021 21:10

English, 09.11.2021 21:10

English, 09.11.2021 21:10


English, 09.11.2021 21:10

Biology, 09.11.2021 21:10

Biology, 09.11.2021 21:10

Mathematics, 09.11.2021 21:10


English, 09.11.2021 21:10