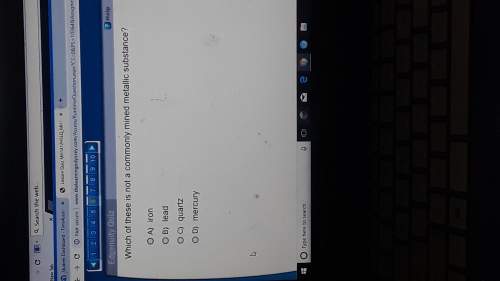
How many moles of Al can be produced from 10.87 g of Ag?
Al (No3) 3(s) + 3Ag -> Al + 3...

Chemistry, 02.04.2020 10:22 aperez4102
How many moles of Al can be produced from 10.87 g of Ag?
Al (No3) 3(s) + 3Ag -> Al + 3AgNo3


Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2


Chemistry, 21.06.2019 23:30
Write a paragraph that provides examples of each stage of volcanic activity, a description of the volcano, and facts about each stage.
Answers: 1

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2
You know the right answer?
Questions



SAT, 02.04.2022 14:00

Chemistry, 02.04.2022 14:00

Biology, 02.04.2022 14:00



Social Studies, 02.04.2022 14:00





Biology, 02.04.2022 14:00

Biology, 02.04.2022 14:00

Mathematics, 02.04.2022 14:00





Social Studies, 02.04.2022 14:00