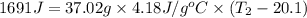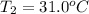Chemistry, 02.04.2020 01:26 kevinkingpin
What is the final temperature of the solution formed when 1.52 g of NaOH is added to 35.5 g of water at 20.1 °C in a calorimeter? NaOH (s) → Na+ (aq) + OH– (aq) ∆H = -44.5 kJ/mol

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 18:00
The human activities in two locations are described below: location a: rampant use of plastic containers location b: excessive use of pesticides and fertilizers which statement is most likely true? location a will have poor air quality because plastic is biodegradable. location a will experience water scarcity because plastic absorbs moisture. the population of honeybees will increase in location b because production of crops will increase. the population of fish in location b will decrease because the water is contaminated.
Answers: 1

Chemistry, 22.06.2019 22:30
Astudent pours 10.0 g of salt into a container of water and observes the amount of time it takes for the salt to dissolve. she then repeats the process using the same amounts of salt and water but this time she slowly stirs the mixture while it is dissolving. the student performs the experiment one more time but this time she stirs the mixture rapidly. the dependent variable in this experiment is: time for salt to dissolve speed of stirring amount of water mass of salt
Answers: 1

Chemistry, 23.06.2019 00:30
What is the percent by mass of magnesium sulfate in mgso4.7h2o
Answers: 3

Chemistry, 23.06.2019 01:30
The solubility of barium nitrate is 9.02 g/100 g h2o at 20°c. a 15.2 g sample of barium nitrate is added to 200.0 g of water at 20°c. is the solution saturated, unsaturated, or supersaturated? a. unsaturated b. saturated c. supersaturated
Answers: 1
You know the right answer?
What is the final temperature of the solution formed when 1.52 g of NaOH is added to 35.5 g of water...
Questions





Social Studies, 17.09.2019 01:30

Biology, 17.09.2019 01:30


Mathematics, 17.09.2019 01:30

Chemistry, 17.09.2019 01:30

Biology, 17.09.2019 01:30

Mathematics, 17.09.2019 01:30

Physics, 17.09.2019 01:30

Mathematics, 17.09.2019 01:30

Mathematics, 17.09.2019 01:30

Physics, 17.09.2019 01:30




History, 17.09.2019 01:30

Physics, 17.09.2019 01:30

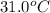
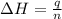
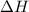 = enthalpy change = -44.5 kJ/mol
= enthalpy change = -44.5 kJ/mol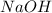 = 1.52 g
= 1.52 g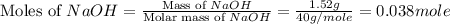
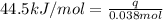
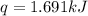
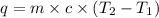
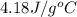
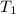 = initial temperature =
= initial temperature = 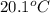
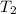 = final temperature = ?
= final temperature = ?