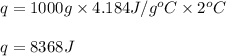Chemistry, 02.04.2020 02:48 maddiefoley17
Suppose you place 125 g of aluminum in a calorimeter with 1,000 g of water. The water changes temperature by 2 °C and the aluminum changes temperature by –74.95 °C.
Water has a known specific heat capacity of 4.184 J/g °C. Use the specific heat equation to find out how much heat energy the water gained (q).

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 08:00
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1

Chemistry, 22.06.2019 17:30
What type of organic molecule comprises the majority of a potato?
Answers: 1

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1
You know the right answer?
Suppose you place 125 g of aluminum in a calorimeter with 1,000 g of water. The water changes temper...
Questions


Mathematics, 29.01.2020 07:42

English, 29.01.2020 07:42





History, 29.01.2020 07:42


English, 29.01.2020 07:42





Business, 29.01.2020 07:42

Chemistry, 29.01.2020 07:42

Mathematics, 29.01.2020 07:42

Mathematics, 29.01.2020 07:42

Mathematics, 29.01.2020 07:42

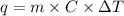
 = change in temperature = 2°C
= change in temperature = 2°C