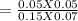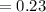An aqueous mixture of phenol and ammonia has initial concentrations of 0.200 M C6H5OH(aq) and 0.120 M NH3(aq). At equilibrium, the C6H5O–(aq) concentration is 0.050 M. Calculate the equilibrium constant, K, for the reaction below.
C6H5OH(aq) + NH3(aq) C6H5O– + NH4+(aq)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
What does x represent in the formula for the compound xcl4?
Answers: 1

Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2


Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2
You know the right answer?
An aqueous mixture of phenol and ammonia has initial concentrations of 0.200 M C6H5OH(aq) and 0.120...
Questions




Chemistry, 17.03.2020 17:29

Mathematics, 17.03.2020 17:29

History, 17.03.2020 17:29

English, 17.03.2020 17:29




Mathematics, 17.03.2020 17:29






Physics, 17.03.2020 17:30

Physics, 17.03.2020 17:30


Mathematics, 17.03.2020 17:30

![K_{c} =\frac{[C_{6}H_{5}O^{-} ][NH_{4}^{+} ]}{[C_{6}H_{5}OH ][NH_{3} ]}](/tpl/images/0577/3457/a8da2.png)