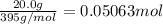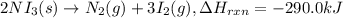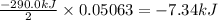Chemistry, 02.04.2020 01:13 wichserc3399
The decomposition of NI3 to form N2 and I2 releases −290.0 kJ of energy. The reaction can be represented as 2NI3(s)→N2(g)+3I2(g), ΔHrxn=−290.0 kJ Find the change in enthaply when 20.0 g of NI3 decomposes.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 12:00
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2

Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1

Chemistry, 23.06.2019 04:00
Calculate the mass of 0.750 mol of the following substance. na3po4. , i'm not quite sure on how to set up the problem to solve! : (
Answers: 1
You know the right answer?
The decomposition of NI3 to form N2 and I2 releases −290.0 kJ of energy. The reaction can be represe...
Questions


Computers and Technology, 14.04.2021 02:40

Mathematics, 14.04.2021 02:40


Geography, 14.04.2021 02:40

Mathematics, 14.04.2021 02:40

Computers and Technology, 14.04.2021 02:40




Mathematics, 14.04.2021 02:40





Mathematics, 14.04.2021 02:40


Biology, 14.04.2021 02:40